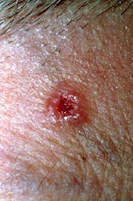
Skin Biopsy: Lesions and Inflammation
Dr Jenny Grew
Biopsies of the skin are generally used to diagnose skin pathologies, but occasionally are used in treatment. This information sheet serves to outline the techniques of biopsy, including: excision (elliptical, saucerisation/ shave, punch), incision, superficial shave, punch and wedge. The type of biopsy a clinician uses is dependent on the pathology of the individual patient.
Non-Pigmented Discrete Lesions
- A biopsy should be performed when a diagnosis is uncertain.
- Complete excision is recommended for suspected malignancy.
- Small and/or nonsuspicious lesions can be removed using punch or shave biopsy.
Excision Biopsy: A well-differentiated squamous cell carcinoma or keratoacanthomas is diagnosed by excising the entire lesion. Partial biopsy often does not present enough information to make an accurate diagnosis.
For a patient with a broad lesion who is prone to keloid/ hypertrophic scar formation, saucerisation could aid in negating surgical and/or cosmetic difficulties, and can be used for the complete removal of in situ/thin lesions. The margins of these samples must remain intact and flat, enabling assessment of the entire sample (achieved by flattening the sample on a card). If atypical/pigmented areas are seen at the base following saucerisation, punch or elliptical excision of the base area may be required to remove further pathology.
Punch biopsy is preferable to shave biopsies, which are prone to sample errors. Many clinicians prefer to use a punch biopsy technique for thick, keratotic lesions. Large, deep lesions require an incisional or wedge biopsy, which includes tumor/fat margins, for accurate diagnosis.
Pigmented Lesions
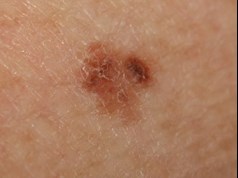
Suspected melanoma:
- Excision margins 1-3 mm
- Small or superficial biopsies may not be representative
- subsequent tumor staging difficult
Partial biopsies can be used:
- If melanoma is unlikely
- For benign, pigmented melanotic macules on the genitals
- Cosmetic concerns
- Broad lesions
- Surgically difficult sites
Saucerisation (including a 0.5-1.0 mm margin) can be performed for ‘ugly duckling’ dysplastic naevi.
If partial biopsy of suspected melanoma is unavoidable:
- The thickest area/s should be biopsied at greater depth
- Multiple small shaves across different areas of a lentigo-maligna-type lesions are recommended.
Small suspicious acral lesions can be removed using saucerisation which include a margin of healthy skin.
Rashes/Inflammatory Conditions
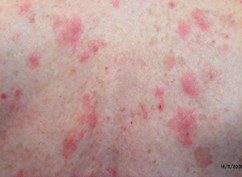
- 4 mm punch biopsy
- Incisional biopsy for larger, deeper lesions
- Saucerisation to remove entire blisters
Many rashes have similar histological patterns. A clinical description of the rash should accompany the biopsy sample and include: distribution, duration and course, other history (e.g. medication), clinical photographs (sent via email or text message) and clinical diagnosis to ensure accurate categorization of the sample.
Stating the biopsy type and reason for the biopsy will ensure that the sample is handled appropriately.
Lesions: A small rim of perilesional skin should be included in a biopsy of tissue that has not been excoriated or ulcerated. Frictional sites and lower limbs may have secondary features resulting in false negative results, and biopsy of these areas should be avoided. Multiple biopsies of the rash and a concurrent biopsy of normal skin may be helpful in disorders that are of polymorphous and pigmented appearance, respectively.
Blisters: Biopsies maintaining the attachment of the blister roof are important. A second biopsy of a non-blistered lesion or normal skin immediately adjacent to the lesion should be taken for direct immunofluorescence microscopy (DIF) and preserved in immunofluorescence transport medium or phosphate buffer saline.
Annular lesions (e.g. porokeratosis): Incisional biopsies should be taken from the center of the lesion in an outwards direction and include a 1 mm margin of normal skin.
Lupus erythematosus, dermatomyositis and vasculitis: The biopsy site is crucial. For lupus and dermatomyositis, biopsy an established lesion (>6 months old) that is active for routine histology and DIF. For vasculitis, biopsy an established, purpuric lesion (>72 hours old) for routine histology and an acute lesion (<24 hours old) for DIF.
Panniculitis: A deep or incisional biopsy, including subcutaneous fat, is recommended. If necrosis is present, a sample should be sent for culture.
Alopecia
- Two punch biopsies
- 4 mm diameter
- Parallel to the direction of hair growth/emergence
Alopecia: Two biopsies should include the entire follicular unit, subcutaneous fat and be at least 5-6 mm in depth (no hairs emerging from the deep aspect). Such biopsies will allow the pathological examination of both vertical and horizontal sections of the biopsy. Clinical information including area of scalp involved, race, duration and clinical differential diagnosis are important.
Biopsy site
- Non-cicatricial alopecia: Most prominent area of hair loss
- Cicatricial alopecia: Area of hair loss but not completely bald (active area at least 6 months duration). Dermoscopy help identify suitable biopsy sites, e.g. areas of scaling or abnormal pigmentation. A second biopsy from the same area can be sent for DIF.
About the Author: Dr Jenny Grew

Anatomical Pathologist
MBChB, FRCPA, AFRACMA
Email: jenny.grew @clinicallabs.com.au
Phone: 1300 367 674
Location: Subiaco, Western Australia
Breast, Gastrointestinal, Molecular Pathology, Cytology, Gynaecological & Cutaneous Pathology
Dr Jenny Grew joined our team at Australian Clinical Labs as Clinical Director Anatomical Pathology for WA in 2017. A graduate of the University of Otago (MBChB, 1992) Jenny began pathology training at Christchurch Hospital (NZ), gaining RCPA Fellowship in 2001. Jenny moved from New Zealand with her family (husband Keith and son Dominic) to Queensland in 2007, taking up the role of Pathologist in charge, providing service to 6 public and private hospitals.